ARTICLE
Weasels, minks and fishers—orphan rehabilitation of the slinky carnivores, Part 2: care protocols for orphaned infants and Juveniles
Peggy Popp
Independent Wildlife Rehabilitator, Mount Horeb, Wisconsin, United States
Abstract
Mustelids are highly specialized, efficient predators with special care needs while in a rehabilitation setting. The following intake and orphan care protocols for select North American mustelids is a combination of natural history, literature review, established orphaned wildlife rehabilitation procedures, professional consultations and personal experience. The protocols address some of the unique characteristics and requirements of these slinky carnivores, while also providing tools necessary for successful rehabilitation and release. Part 1: Natural History (Popp 2024) discusses the natural history of North American weasels, minks and fishers, including physical characteristics, habitat and diet, and how these factors may be used to inform their intake and care protocols when brought into the rehabilitation setting. Part 2: Care Protocols for Orphaned Infants and Juveniles discusses vetted care protocols for orphaned weasels, minks and fishers based on their natural history and established orphaned wildlife rehabilitation procedures. Part 3: Pre-Release Conditioning and Release discusses the steps involved in preparing these species for a successful release.
BIO
Peggy Popp holds an Advanced License for wildlife rehabilitation in Wisconsin, with Endangered and Threatened Species Authorization. She has been an independent home‑based wildlife rehabilitator since the mid‑1980s, working with orphaned small mammals. Since the summer of 2013, when an infant least weasel appeared on her doorstep, she has focused her attention on raising orphaned mustelids, researching ways to improve care, and promoting best practices for the rehabilitation of mustelids, especially those found in Wisconsin: the three species of weasels (least, short-tailed, and long-tailed), minks, and fishers.
Keywords
Mustelid; weasel; mink; fisher; carnivore; orphan mammal
Abbreviations
ADV: Aleutian mink disease virus
GI: gastrointestinal
REM: Rapid Eye Movement
Citation: Wildlife Rehabilitation Bulletin 2025, 43(1), 22–37, http://dx.doi.org/10.53607/wrb.v43.304
Copyright: Wildlife Rehabilitation Bulletin 2025. © 2025 Peggy Popp. This is an Open Access article distributed under the terms of the Creative Commons Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/), permitting all non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Accepted: 21 June 2025; Published: 13 August 2025
Competing interests and funding: The author has nothing to disclose.
This work was not supported by any funding agencies.
Correspondence: Email: peggy2.423@gmail.com
Introduction
Combining a knowledge of natural history, physiology, established orphaned wildlife rehabilitation procedures and personal experience, along with literature reviews and consultations with experts, these protocols for raising orphans of select North American mustelid species address their unique characteristics and requirements. The discussion also highlights key aspects necessary for a successful rehabilitation and release back into the wild.
This paper focuses on how the natural history informs the care protocols for the rehabilitation of orphaned fishers, minks and the three North American species of weasels.
As discussed in Part 1: Natural History (Popp 2024), weasels, minks and fishers are obligate carnivores with a high metabolic rate. They have relatively simple stomachs and short GI tracts with no special adaptations for digesting plant matter, resulting in a GI transit time of only 3–4 hr. Combined with their high metabolism, they cannot survive long without food and the smaller mustelids, in particular the least and short-tailed weasels, must eat as often as every 2–3 hr throughout the 24-hr cycle.
Mustelids are susceptible to human toxins such as rodenticides and lead from both the environment and their prey (Hornshaw et al. 1983; Blus et al. 1987; Luson et al. 2014 Llungvall et al. 2017), as well as respiratory viruses such as SARS-CoV-2, and avian influenza A(H5N1) virus (Taylor 2014; Molenaar et al. 2020; Agüero et al. 2022). Biosecurity precautions in rehabilitation facilities must be taken to protect both the animals and the caretakers, and masks, gloves and air filtration units are recommended during outbreaks of human respiratory illnesses such colds, influenza and COVID-19.
Most mustelids delay implantation during reproduction; the delay can range from almost a year (fishers) to a few weeks (minks), and is followed by a short (3–7 week) active pregnancy. An exception is the least weasels, who do not delay implantation, usually giving birth within 35 days of breeding (Heidt 1970). The young are born in the spring and generally begin eating meat soon after their teeth emerge, which may be before their eyes open, but they are not weaned until later. They remain with their family units over the summer learning to hunt, and disperse in late summer (minks and the weasels) or fall (fishers) (Aubry et al. 2005). All remain in their natal home ranges in the early stages of becoming independent and dispersing, before venturing further to establish their own territories. The mother offers a protected territory and may assist in providing food for her offspring as they become independent; she essentially provides a soft release for her young. Least weasels may be born at any time of the year (Hall 2015), but most often in the spring and late summer or early fall.
Habitat preferences vary within and among mustelid species, all of which except sea otters are basically terrestrial (living on the surface of the land). Minks are also semi-aquatic; fishers are semi‑arboreal, often inhabiting and climbing trees and the weasels are also semi-fossorial, spending much of their time underground, as well as semi-arboreal. Young are born in dens or deep in a tree cavity. To improve welfare and reduce stress while in the rehabilitation setting, the habitat in captivity should mimic the natural habitat appropriate to the age of the mustelid in care.
Comparison charts with species-specific details are available in Appendix A. Species-specific orphan protocols are available in Appendix B.
Initial response
Mustelid orphans are an exception to the “watch and wait” rule. They nest in dens and anytime an infant is found outside of the den with no parent in sight it needs help, and it needs help as quickly as possible due to its high metabolism. Juveniles spend the summer in their family units under the watchful eye of their mothers, learning how to hunt and maneuver through their environments safely, returning to the den site to rest. Juveniles found alone or huddled with a sibling, relatively easy to capture and without a parent nearby, need assistance and again, due to their high metabolism, they need it quickly.
Very young fisher and weasel infants allow their body temperatures to cool and go into a slight torpor when their mothers are away from the den, to conserve energy (King & Powell 2007a). This may also be true of minks. When the mother returns, she warms the kits and they quickly become active and begin to eat. Cool and even cold kits coming into care should be warmed and then fed as soon as they begin to get active. Once warmed, infants in care must be fed frequently.
Because they are so long and lean, and because their fur is so rich and dense, there may not be much difference in the appearance of a healthy wild mustelid and a starving mustelid. They should be isolated in warm, dark and quiet locations and, once warmed, should be immediately fed. Because of their rapid GI transit time and need for calories, rehydration is combined with an easy to digest protein and fat.
Using water or oral rehydrating solution as the diluent, neonates, infants and young kits not yet old enough to be weaned are started with over-diluted formula (10–20% full strength) while older kits and adults are given diluted (25–50% prepared full strength) EmerAid® IC Carnivore (EmerAid, Cornell, IL), Oxbow Critical Care Carnivore® (Oxbow Animal Health, Omaha, NE) or chicken in broth (human) baby food (Beech-Nut®, Amsterdam, NY), which provide easy-to-digest protein. Rather than using the traditional approach of flushing out the infant GI tract with feedings of an oral rehydrating solution, such as lactated Ringer’s solution or normal saline, use the rehydrating solution or water to provide diluted food as soon as possible. Feed a kit, slowly, as much as it wants and at frequent (20–40 min) intervals. Food can initially be offered with a syringe as necessary and older, eyes-open kits will often be capable of self-feeding in a relatively short time. The feedings can be repeated with gradually less dilution and at longer intervals as the kit consumes the food and begins to perk up. Monitoring needs to be done for adverse effects (vomiting, deteriorating condition, not touching the food, etc.). Juveniles should be left with a shallow dish of the food in the cage for at-will consumption. This process will rehydrate the kit while also supplying critical protein and calories. Frequent small meals, and at-will consumption, are key. Maple syrup on the gums can be used to counter hypoglycemia, with less of a blood sugar peak and crash compared to corn syrup (St-Pierre et al. 2014). Kits are often on full strength formula or a regular diet within 12–36 hr, depending on their condition at admission.
Infant weasels and minks may have swollen genitalia, which is sometimes bulbous and red (Figs. 1 and 2). This is a natural response to the hormonal surge of newborns; the swelling will subside on its own and typically no intervention is necessary.

Fig. 1 Short-tailed weasel infants with a characteristic mane, and the swollen genital area. Photo credit: Valeria Villazon.

Fig. 2 Swollen genital area of an infant female mink.
Medical considerations
Heightened biosecurity precautions need to be taken with all mustelids. They are susceptible to many human respiratory illnesses, including COVID/SARS-CoV-2 and influenza, as well as avian influenza and canine distemper (Devaux et al. 2021; Zhou & Shi 2021). If mink fur farms or ranches are in the area, wildlife may have been exposed to ADV (Mayer et al. 2015). It is important to be aware of disease risks in one’s area and follow current biosecurity recommendations. Note that disinfectants must be thoroughly rinsed to remove residues, due to the mustelid scent sensitivity.
Medication and vaccinations should follow ferret guidelines, and if ferret protocols are not available use cat or kitten guidelines. Consult with a veterinarian trained in treating ferrets or other mustelids before using any vaccine not specifically authorized for mustelids. Be alert for indications of stress-induced ulcers, which may be treated with stomach protectants. Note that body weights may vary greatly depending on how recently a kit last ate or defecated.
Internal and external parasites may be found in or on any mammal but are not found often enough with mustelids to warrant routine precautionary treatment. This may be due to a combination of their dense fur and rapid GI transit time. If internal parasites are suspected due to bloating, not gaining weight, diarrhea or stools with blood or mucus, a fecal examination should be performed to determine if a parasite is the cause, and treatment should be conducted accordingly.
Additional medical management can be found in Chapter 22, Natural History and Medical Management of Mustelids, of Medical Management of Wildlife Species: A Guide for Practitioners, and Chapter 48, Mustelidae, of Fowler’s Zoo and Wild Animal Medicine.
Taurine
While many animals, including humans, produce taurine, mustelids (and felids) must get taurine, an essential amino acid, from their food, as their body cannot produce it. Muscle meat is high in taurine, as are spiders, many insects, brains and eyes. Chicken baby food, chicken dark meat, clams and clam juice, tongue and heart are also good sources of taurine. Taurine is present at particularly high levels in the milk of obligate carnivores; however, taurine is not always added to infant formulas. Kitten and Zoologic® Milk Matrix (Pet-Ag, Hampshire, IL) formulas are recommended because they contain supplemental taurine; Esbilac® Puppy Milk Replacer (Pet-Ag, Hampshire, IL) does not. Most Fox Valley™ Day One (Fox Valley Animal Nutrition, Lake Zurich, IL) milk replacer powders (FV) have supplemental taurine added to the formulation. Formula ingredient labels should be carefully reviewed prior to use, and, due to formulation changes, periodically re-checked.
While the specific mustelid requirement for taurine is currently unknown, milk formulas which do not include taurine-supplemented formulas as a base should be supplemented with taurine powder. The recommended taurine supplementation for felids is 250 mg/animal/day (AZA 2010a). For striped skunks, a balanced diet should be supplemented with chewable taurine tablets (250 mg juveniles, 500 mg adults/per day) (Abou-Madi 2019). Taurine is not considered to be toxic, with no known adverse effects even at high doses, indicating it is safe to supplement at 250 mg/animal/day. Taurine tablets are sold as a human supplement, which may be ground to a powder and added to the mustelid formula and food.
Stress
Mustelids may be susceptible to capture myopathy and are considered high stress species when in captivity (Frost & Krohn 1994). They fuel their high metabolic rates through hunting, and they hunt by responding rapidly to stimuli such as light, sound, smells, vibrations and movement in their environments. Exposure to a barrage of unfamiliar light, sound, smells, vibrations and movement in the rehabilitation setting will increase the stress of an already stressed animal. Signs of stress include “poofing” (discharge of anal glands), vocalization (e.g., squeaks, growls, screams and shrill cries) and fear behaviours (cowering, hiding, backing away and fear aggression). Infants, with closed eyes and ears, rely primarily on their sense of smell and touch, and should be cared for by a limited number of caretakers to reduce stress. A dedicated pair of cotton gloves worn over disposable gloves and a dedicated oversized shirt or scrub top worn over regular clothing, which are left on top of the container in between feedings, can help mask a rotating group of caretakers. The use of scented products, including scented laundry and hand soaps, should be avoided.
With their high stress factor and need for a whole prey strict carnivore diet, appropriate pre-release conditioning and an extended supported release (similar to hacking), the mustelids may have more in common with raptor rehabilitation protocols than they do with more common small mammals coming into care such as squirrels and raccoons.
Single orphans
As small predators, mustelids are in a predator/prey relationship with most species, including other mustelid species. They tend to live at low densities and come into rehabilitation rarely and irregularly. Different species of mustelids, including different species of weasels, must not be combined due to their predator/prey relationship. While raising a single animal is never desired, sometimes a single animal arrives and none others are available to provide a foster sibling. While not ideal, raising a single orphan for a successful release is manageable and acceptable. The caretaker will need to invest more resources into meeting the needs of the young animal to ensure security, adequate conditioning and education and preparation for release. Even young infants coming into care and raised as singles will recognize others of their species as kindred, while other animals are usually seen as prey or predator. The challenge is to first provide sufficient security for the orphan while limiting exposure to other humans and potential predators, and, as they get older, to adequately stimulate and prepare them with the necessary life skills for a successful release.
Singles of the same species and similar developmental age can be grouped together, and often it is possible to put a single of the same species and similar developmental age with a sibling pair. It is important to be careful to avoid overcrowding, which will certainly result in stress and may result in aggression and injuries. It is important to match developmental ages because stronger, more agile animals may bully, and perhaps injure or kill, weaker or less agile ones. Conversely, the larger may “adopt” the smaller ones and they may become special buddies. Generally, matching strength and coordination is more important than matching age and size. Conflict seems to occur more often between mismatched males than between male/female or female/female pairs. Mustelids play rough, which is excellent training for the real world, but if indications of significant stress appear during interactions (screaming, fear pooping, “poofing” or injury) animals must be separated. If that results in singles, it is unfortunate but still necessary to protect weaker animals. Aggression between cage-mates can happen with natural siblings as well as with foster siblings, especially if overcrowded, in too small of a space or lacking sufficient enrichment. If separation is necessary, they can be housed in adjacent units to maintain same-species exposure. Sometimes they can be re-joined in the pre-release enclosure or during supervised playtime, but this will require careful monitoring.
If a single orphan is in care it is important to provide them with a soft heartbeat comfort toy, such as a Snuggle Puppy® (SmartPetLove, Novi, MI), to reduce stress, and to provide opportunities to chase, tussle and interact, all of which they would get with a sibling. A trusted caretaker (and all mustelids should have a minimum number of designated caretakers) can use a cat wand to provide chase and tussle opportunities, fasten a soft toy attached to a bungee on the side of the enclosure, throw toys around for chasing and provide young juveniles with supervised activity time.
Infant care—feeding
Mustelid milk is high in protein (26–40%), high in fat (31–45%) and low in carbohydrates (8–21%) (AZA 2010a). Smaller species of mustelids require higher levels of fat and protein and lower levels of carbohydrates than larger mustelid species. Minks also require supplemental fish oil, and fishers require relatively less protein and fat but more carbohydrates than the smaller mustelid species. Several vetted options exist among formula powders, which is important given the inconsistent availability and formulation changes of commercial products.
Weasel and mink formula is a combination of two-parts Fox Valley (FV) Skunk (32/45), which has been reconstituted per the package directions, to one-part KMR® Kitten Milk Replacer (Pet-Ag, Hampshire, IL), which has also been reconstituted per package directions. Weasel formula should also have 1–2 teaspoons Milk Matrix 30/52 powder per cup of prepared formula to boost the fat content. Fisher formula is Milk Matrix 33/40, which is a low lactose formula with taurine, or a 1:1 combination of KMR (reconstituted at one part powder:three parts water) and Milk Matrix 33/40 (reconstituted at one part powder:two parts water). If Esbilac® (regular, or Goat Milk Esbilac® (GME)) (Pet-Ag, Hampshire, IL), is used for fishers it must be supplemented with taurine; Esbilac is not recommended for weasels or minks.
Infant mustelids are often lactose intolerant, which can be fatal if not managed promptly. Infant lactase drops, which are not prepared with alcohol, should be available for all mustelid infants should there be indications of lactose intolerance, such as bloating and decreased appetite.
Wombaroo© (Glen Osmond, South Australia) Otter (30/50) is an alternative for Milk Matrix 30/52; both are low lactose, nutritionally complete formula powders, and contain taurine. FV Beaver (30/50) is also nutritionally complete and similar in the protein to fat ratio, but contains added lactose. FV Ultra Boost (25/40) is a high fat creamer additive, but is not nutritionally complete.
Milk Matrix 42/25 and Wombaroo Cat (41/29) are equivalent to PetAg KMR (40/28) in the protein to fat ratio and all are supplemented with taurine. The Milk Matrix and Wombaroo products are formulated to be low lactose, while KMR is supplemented with Bene-Bac® (Pet-Ag, Hampshire, IL) probiotics. Milk Matrix and PetAg formula powders tend to be slightly higher in protein and fat than the equivalent Fox Valley or Wombaroo formula powders; Milk Matrix and Wombaroo formula powders are generally formulated to be low lactose, while most Fox Vally formula powders contain added lactose. Esbilac and KMR formulas are manufactured for the domestic pet market and formulations may change to better meet the needs of dog and cat owners, while Milk Matrix, Wombaroo and FV are formulated specifically for wildlife. The ratios provided above are based on pre-mixed, liquid formulas; powdered formula ratios are provided in a chart format in the supplemental Orphan Care Protocols (Appendix B). Note that FV Skunk directions mix two-parts powder to three-parts water, while the other powders are generally mixed one-part powder to two-parts water. If KMR is used to prepare fisher kit formula, it is mixed at one-part powder to three-parts water.
A 0.5 mL syringe fitted with a Mini Miracle Nipple® (Miracle Brand, Somers, CT), or equivalent, is recommended for feeding weasel infants, and a 0.5 mL, 1 mL or 3 mL syringe fitted with a Mini Miracle Nipple is recommended for mink infants. A 10- or 20-mL syringe fitted with a Small Puppy Miracle Nipple® or the Easy-Grip Nipple (Henry’s Healthy Pet Foods, Inc., Floyd, VA), or a small baby bottle fitted with a size 0 (very slow flow) nipple (e.g., Dr. Brown’s Natural Flow® Anti-Colic Options+™ Narrow Baby Bottle, 2 oz/60 mL, with Preemie Flow Nipple, Handi-Craft Company, St. Louis, MO), may be used for infant fishers. If the infant spits the formula up after eating, the formula may have been given too fast. Use a smaller syringe or allow brief rest intervals while feeding. To encourage an infant fisher to latch onto the nipple, it may be beneficial to first dip a gloved finger into the formula and allow the infant to latch onto the fingertip before sliding the nipple in alongside the finger, then withdrawing the finger (Figs. 3 and 4). In the author’s experience, weasel and mink infants do not have a strong sucking reflex and the formula is slowly injected through the nipple using a syringe.
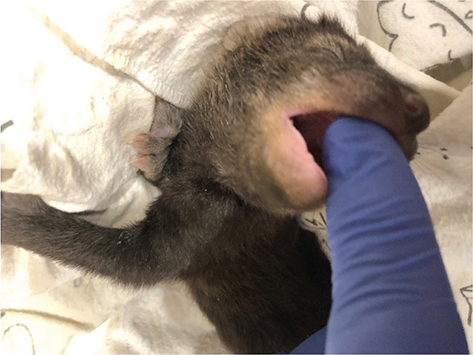
Fig. 3 Infant fisher latching onto a fingertip. Photo credit: Elliot Lozano.

Fig. 4 Feeding an infant fisher with a syringe and nipple. Photo credit: Emerson Paton.
A note about mustelid teeth: the appearance of the canines is often the first indication that the teeth are emerging, signalling a readiness to begin eating solids. At around the same time, the carnassial teeth are also emerging. The carnassial teeth are the shearing teeth made up of the fourth upper premolar and the first lower molar, working in tandem like kitchen shears to enable slicing through meat and cutting tough sinews. If the infant mustelid is trying to chew on the nipple from the side of its mouth, it is working its carnassial teeth to slice the tip off the nipple. This is often highly effective and can destroy expensive nipples in a short time, and there is also a risk of the nipple tip being swallowed. The chewing on the side of the mouth while syringe feeding should be discouraged! If they do slice the tip off, do not panic but do watch for potential GI blockage from a swallowed bit of rubber, and discard damaged nipples. The author has never had an issue with blockage, even when they presumably swallowed the rubber tip, but the caretaker should be alert for symptoms of abdominal blockage. For information on symptoms of blockage, see https://vcahospitals.com/know-your-pet/gastrointestinal-diseases-in-ferrets.
Minks require supplemental fish oil, such as salmon oil (e.g., Grizzly Salmon Plus™, Woodinville, WA) at all stages of development. Infants can be given 0.1–0.3 mL oil supplement directly into their mouth at feeding time, and 0.3–0.5 mL topping off dishes of meat. Start with every feeding or every other, adjusting the amount or frequency if flaking skin is noticed, especially on the neck and shoulder area (Fig. 5). Such skin flakes are common with orphaned infant minks, and may also be due to their rapid growth. Olive oil should be rubbed onto affected areas to combat peeling and flaking. Personal experience has demonstrated that olive oil outperforms avocado oil, and coconut oil is ineffective as a treatment for flaking skin. Mink guard hairs are quite oily, which renders the pelage water-resistant; the author believes that in the den the mink kits are regularly “oiled” as they crawl over and under their mother, and the kits coming into care quite young (<3 weeks old), before their guard hairs appear, often require an external application of olive oil on a regular basis, as well as the fish oil dietary supplement, to combat dry and flaking skin.
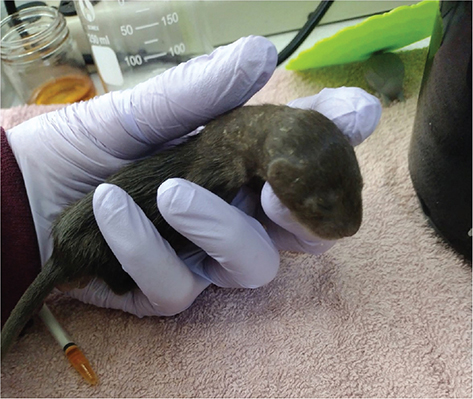
Fig. 5 Infant minks often develop flaking skin on their neck and between their shoulder blades. Regular application of olive oil will help to alleviate the flaking.
Infants must be stimulated to urinate and defecate at each feeding until reliably doing so on their own, which is usually by the time their eyes open but often before. The orphan, especially minks, may object—loudly—to this procedure, and may even “poof” the first few times. They may also fuss when eliminating on their own. If the kit is constipated, the formula may be diluted up to 25% with water or another rehydrating liquid; this may require additional feedings to ensure adequate nutrition.
Once stabilized and on full strength formula, feed infants about every 2–4 hr, around the clock. Larger species will prefer longer intervals than smaller species, and neonate least or short-tailed weasels may need to be fed more often. Signs of hypoglycemia (cool, lethargic) indicate the interval may have been too long; warm the infant before feeding and decrease the feeding interval. Feed as much (or as little) as each animal wants at each feeding; the amount may vary considerably from one feeding or one infant, to the next, but it is often in the 2–5% body weight range for minks and weasels and 3–10% body weight range for fishers (P. Popp 2019 and E. Lozano 2024. Personal communication of unpublished data.). Weasels and minks will become slightly more mobile and have their canine and carnassial teeth erupt at 2–4 weeks, a few weeks prior to their eyes opening, at which point they may start sucking on meat placed in a small, shallow jar lid. Fishers will also become more mobile prior to their eyes opening, but they begin eating meat around the time their eyes open, at about 7 weeks.
This is a lot of infant feedings every day! One approach is to set up 24 hours of phone alarms for each feeding at the start of the day. To increase efficiency, a baby bottle or baby food warmer (e.g., the Easi-Warm Electric Bottle and Food Pouch Warmer, Tommee Tippee, Stamford, CT) may be used as a water bath to reduce the preparation time. The Tommee Tippee bottle and food warmer is recommended for temperature control, reliability and versatility. Filled with distilled water, and replacing the water every day or two (or sooner if contaminated) it can be left on throughout the day and night at the appropriate temperature; a silicon cup lid on top will reduce evaporation and heat loss between feedings (Fig. 6). The water should be slightly warm to the touch. When syringe feeding, a small glass jar (baby food, spice jar, etc.) inside the bottle warmer can be filled with tap or distilled water and the formula container placed in the jar to warm, thus preventing contamination of the water in the bottle warmer unit. The inner jar can be easily removed for cleaning after each feeding, refilled with fresh water and put back in the warmer to be warmed and ready for the next feeding.
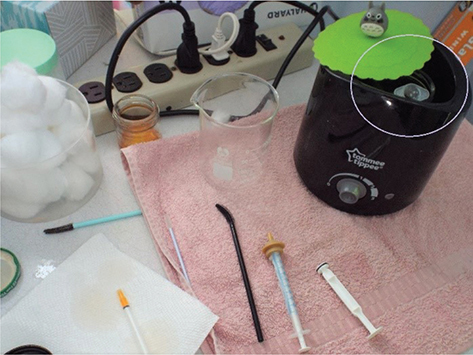
Fig. 6 A baby bottle or baby food warmer may be used as a water bath, with a small, water filled jar inside used to hold the formula container necessary for the feeding.
For syringe feeding, prepare a small container large enough to hold the formula needed for the feeding (often a 3-, 5- or 10-mL syringe, or a 1 oz Tupperware “midget” can be used). The formula goes in the small container or, if using syringes, the syringes are partially filled and capped, the plunger removed and the syringe placed in the jar (Fig. 7). Do not overfill, or the formula will overflow into the water bath when the feeding syringe is inserted to withdraw the formula.

Fig. 7 At feeding time, a small container large enough to hold the formula needed for the feeding goes in a small jar or syringe. If using syringes to warm the formula, the syringes are partially filled and capped, the plunger removed and the syringe placed in the jar.
This system minimizes waste, warms the formula quickly and keeps it at the appropriate temperature for the duration of the feeding. The warm water can also be used to dampen a Q-tip or cotton ball for stimulating the infant or for clean-up. After each feeding the leftover warmed formula is discarded, and the small formula container, feeding syringe or bottle and nipple are rinsed, then refrigerated and re-used for the next feeding. Feeding supplies are separate for each cohort, and replaced every 24 hr; re-using them in this way cuts down dramatically on the number of syringes, bottles and nipples that need to be used (and washed). The bottle warmer should be rinsed regularly and the water replaced with fresh water. Occasionally filling the warmer unit with a vinegar solution and heating for an hour or so will both clean and descale the apparatus, followed by a rinse and refill with clean distilled water so it is ready for the next feeding.
After the infant has been stabilized and is on full strength formula, a solo caretaker should adjust or delay one feeding during the night, ideally during the latter part of the night, to facilitate getting a block of sleep with a complete REM sleep cycle. In a perfect world, a team of two or three people care for the infants, to trade off night feedings. The feeding schedule is relentless, and sleep deprivation may lead to serious, sometimes fatal, mistakes. The caretaker must also prioritize their own health, to be able to care for the orphans.
The weaning process is very gradual, often starting when their teeth first erupt. Mustelids do not have “milk teeth” so much as “baby meat teeth,” which will be replaced by their permanent teeth as they mature. Initially, the infants sucking on solid food serves to supplement their growing body and insane metabolism, taking some of the burden off their nursing mother and providing the kits with food if she is delayed in returning. She will leave opened prey behind for the kits to suck on while she is out hunting. The neck, front legs and shoulders of an infant mustelid develop first, and those features are quite strong from birth, allowing an infant to hold their head up. By the time teeth begin to emerge, kits are moving around and capable of holding their heads and necks up sufficiently to eat out of a shallow dish. A baby food jar lid works well for weasels and minks, and larger jar lids or very shallow dishes for fishers (Fig. 8). It may be possible to gradually extend the interval between formula feedings once they are self-feeding noticeable amounts of food.

Fig. 8 Introduction to solid food is very gradual, often starting when their teeth first erupt.
The pattern of introducing prey meals is the same for all of these “slinkies” and mimics what their parent would be providing—start the young animals on whole prey, sliced open or chopped up to make the soft tissue accessible. They will suck on the meat, sometimes taking bites. Chopped or ground raw meat of deer, rabbit, rodent or a raw commercial product available for cats consisting of muscle, bone and organs, is an option that can also be used later as a supplement to whole prey. Avoid feeding beef or pork.
Once the infants have teeth and are interested in solid foods, a small portion of the weaning food can be left in their cage between formula feedings. It is important not to reduce the frequency of formula feedings, which is their primary source of sustenance, during this time. Expect formula intake to vary depending on how recently the kit was eating the weaning food. Providing a small amount of solid food (often only a spoonful or so), replaced at frequent intervals to maintain freshness, ensures constant availability with minimal waste. Small dishes of formula and water should also be made available at this time. There is no need to mix formula with the solid food to introduce solids. Soon after their eyes open they may start to resist being handled for syringe feeding, and transition entirely to dish feeding. Jar lids and XS Repti Rock Food and Water Dishes (Zoo Med Laboratories, San Luis Obispo, CA) work very well for this phase. Continue to offer formula, via syringe, bottle or dish (but encouraging the transition to a dish), for as long as they want it. Formula is an excellent source of nutrition, and gradual weaning mimics their natural history. As they rely less on formula it is important to have a shallow dish of clean water available at all times.
Infant care—housing
Infants can be housed in an incubator (80–85 deg F) or (preferably) in an aquarium or crate partially set on a heating pad which has been set to low. It is important for them to be protected from drafts, and also that they not be overheated, which can cause hair loss and even death. Offer nest areas for sleep and rest periods. In addition to a comfort soft toy, fleece pieces flat-stacked into a tower, piled up fur pieces or fleece with flannel snug sacks may be used for bedding material; when placed over the warm side of the container (if using a heating pad) these items make a fine, cozy den. The infants will rearrange or burrow in to find their own temperature comfort range. Lining the bottom of their enclosure with sheets of paper towel makes it is easy to change out the soiled areas and keep the bedding clean. Infants should be transitioned out of an incubator and into an enclosure with a heating pad once they are increasingly active, and before offering dishes of food. Food and formula dishes go on a clear space of the paper towel over the unheated side of the enclosure. The latrine area is generally away from the nest. Soiled paper towel is readily changed out for spot cleaning, or a fresh piece of paper towel can be placed on top of the soiled layer at each feeding and the stack changed out periodically. This arrangement allows the scent of the latrine area to remain while limiting exposure to the soiled area. Mustelid infants prefer to stay under cover; they will come out to eat and to eliminate, usually backing up against the side of the cage or a dish to defecate, before burrowing back into their sleep area (Fig. 9).

Fig. 9 Food and formula dishes go on a clear space of the paper towel over the unheated side of the enclosure (a soft-sided carrier). The single infant weasel is burrowed into the Snuggle Puppy.
A comfort heartbeat unit wrapped in a piece of fleece, or a heartbeat stuffed animal such as a Snuggle Puppy, is often soothing to infant mustelids, and is especially important for a single infant (Fig. 10). Signs of stress may include circling, restlessness and vocalizing.

Fig. 10 A Snuggle Puppy helps to comfort a single young fisher.
Juvenile care—feeding
As their teeth emerge and jaw strength increases, kits will begin tearing off pieces of prey and consuming the chunks. When transitioning from chopped to whole prey, keep in mind the fact that infant and juvenile prey species are easier to eat (softer bones and easier to tear skin) but with a different nutritional profile compared to an adult mouse, rat or rabbit (Kwon et al. 2022).
Mustelids may “imprint” on their food, defined as any kind of phase-sensitive learning (learning occurring at a particular age or a particular life stage) that is rapid and apparently independent of the consequences of behaviour, although there does not seem to be sufficient information to know the time period for mustelid kits. Enough is known, however, to underscore the importance that the kits, both infants and juveniles, be exposed to the natural prey likely to be found in the release area. For weasels, this means furred mice and voles, and perhaps earthworms and insects; for minks it means mice, voles, rats, muskrats and aquatic species (fish, crayfish, frogs) found in their home environments and for fishers it means rabbits and hares, rodents, grouse or turkey poults, venison and porcupines. Fishers can also be offered seasonal foraged native fungi, such as truffles, and wild-harvested berries, such as blueberries, mulberries and black raspberries, or apples. Berries can be offered on the branch or cane, to provide enrichment as well as delicious nutrients. Do not feed commercial fruit to wildlife, as commercial fruits have a much higher sugar content than wild fruits.
A base diet of ground or chopped venison, rodent, rabbit, duck, chicken or quail with organs and bones, or a high protein, taurine-supplemented commercial raw cat food, is a practical, convenient and economical base or supplement, provided the commercial diet is adequately supplemented with whole prey (Fig. 11). Stella and Chewy’s (Oak Creek, WI) makes a fortified frozen raw cat food product with “98% meat, organ, and bone” in convenient small nuggets available at many pet food stores, and companies such as My Pet Carnivore.com (Indianapolis, IN), Top Quality Dog Food.com (Hyattsville, MD) and Raw Feeding Miami (Miami, FL) offer raw grind options with meat, organ and bone for pets, usually with limited local delivery areas and shipping across the United States. An internet search can often find local pet stores carrying frozen raw grinds, preferably from local suppliers. Note that most providers of raw grinds state it should be used as a supplement only, and if fed to cats should be supplemented with additional vitamins, including taurine. If the base diet is an unfortified raw grind, it should be fortified with supplemental nutrients, e.g., the supplement recommended by the Association of Zoos and Aquariums (AZA) (2010b): Mazuri® Carnivore Supplement for Whole Prey, a blend of nutrients designed to complement raw meat diets that contain bones, or Mazuri® Carnivore Supplement for Slab Meat, to supplement raw meat diets without bones (Land O’Lakes, Inc., Arden Hills, Minnesota, USA).

Fig. 11 Raw ground food, topped with salmon oil and vitamins, and supplemented with whole prey, is offered to young minks. Mustelids, unlike rodent species, do not gnaw on the plastic ReptiRock food and water dishes.
Choose options that will mimic natural foods, such as rabbit, venison, muskrat, duck, quail, chicken and fish, avoiding beef and pork, and look for an option made with meat, organ and bones with no cereal or other plant-based additives. Avoid donated meat harvested with lead ammunition or potentially contaminated with rodenticides. My Pet Carnivore carries such items as ground whole prey muskrat that was wild-trapped, whole prey rabbit, whole prey chicken and raw, frozen, whole prey smelt. Fresh water whole smelt provides a convenient fish option for minks, and it may be possible to source frog leg or other aquatic trimmings from a local fish market. If a significant portion of the mink diet is fish, the fish must be supplemented as appropriate, especially with thiamine and vitamin E (e.g., Mazuri). Fishing Cat & Otter Supplement). Dehydrated or freeze-dried raw food must be reconstituted before being offered. A base diet of high-quality raw grind available for free feeding, supplemented with vitamins, along with a regular offering of whole prey, will help ensure the calorie and nutritional needs are met. Providing whole prey not only allows practice opening and eating, and familiarity with, whole prey, but also ensures complete and balanced nutrition (intact whole prey includes the entire GI system, including the small intestine, an important source of partially digested carbohydrates [King & Powell 2007b]). The base diet helps to ensure food is always available to all individuals in the enclosure, while prey items are often immediately removed by an individual and cached, if not eaten.
Continue to provide daily fish oil (in a dish or on their food) for minks. Weasels are unlikely to touch fish oil and fishers are not impressed with it. Also continue to provide formula in a dish for as long as any one animal in the enclosure is consuming it, and provide clean water at all times, in a dish similar to what is used for solid food. Some mustelids might use an appropriately sized water bottle but should also have clean water in a dish.
Juvenile care—housing
Mustelids can be escape artists, with lightning fast movements and the ability to slink through unexpectedly small openings. When it comes to weasel housing, it is important to realize that anything permeable to mice may also be permeable to weasels. Minks are masters at opening cage doors, and, in addition to being very fast, all mustelids are excellent climbers.
Enclosures. As kits become more active they should be moved into larger cages with an increasing variety of enrichment. Balancing the stress of being moved with the need for more space, they can be placed in a small area of their juvenile enclosure and this area expanded as they grow. This practice allows the young to explore and expand their ranges within their comfort zones, and mimics their behaviour in the wild. If initially housed in a small carrier, the infant carrier can be placed inside a larger enclosure, with the door of the carrier opened or removed. For example, allocate a two-story Critter Nation® ([CN] MidWest® Homes for Pets, Muncie, IN) cage, or tandem double two-story CN cage (two two-story cages fastened together, with the adjoining walls removed on one level), with one unit initially set up as a complete space with a sleep, food and latrine area. As they become more active, more areas of the cage can be made available. Alternatively, a canvas “Puppy Playpen,” such as the Frisco© Soft-Sided Dog, Cat & Small Pet Exercise Playpen (Frisco by Chewy, Chewy, Inc., Plantation, Florida), with both a connected top and a connected bottom, can be used to house young juveniles, or to provide a larger play area for active times. Be sure to seal up the “water bottle” openings of a puppy playpen, if present.
Floor pads in the cage unit not only provide an easy to replace and reusable substrate, but also offer hiding and burrowing opportunities (Fig. 12). Avoid using disposable “puppy pads” as the pads are easily torn up and may be ingested or become an entrapment risk. If washable pads are not available, or in addition to washable pads, placing hammocks, t-shirts or sweatshirts or smaller fabric pads, as well as leafy or conifer branches, on the cage floor, offers similar burrowing or hide opportunities.

Fig. 12 Long-tailed weasel burrowed under the paper towel and cage pad.
Continue to use easily replaced paper towel liners under food dishes and in latrine areas. Litter boxes can be used for juvenile minks and fishers, as space allows.
Move young juveniles, along with the current bedding, from their infant housing (or simply move the soft crate or pet carrier and then fasten open the doors) to their new space. Bring over all the soft bedding and enrichment items from the original cage into the new one, and supplement the extra space with fresh bedding and enrichment items (Fig. 13).

Fig. 13 Enclosure for young juvenile weasels, previously housed as infants in the canvas pet carrier on the upper level. The sides and front of the cage unit are hung with sheer curtains, which are generally kept closed.
When it is time to clean, replace only the objects that are soiled, a few of the bedding or enrichment items at one time, rather than a “clean sweep” housecleaning, to minimize stress. The AZA recommends changing no more than 25% of old, soiled furnishings at one time (AZA 2010c). A couple of new things in an otherwise familiar space is exciting, while a completely new environment with an unfamiliar scent is frightening and stressful. Store clean bedding in the same room, on or under the cages, if that is an option, so that it absorbs the “local” odours.
Minks and fishers will often use ferret or rabbit corner or square litter boxes (high back, low access point) for toileting, while weasels generally will not; paper towel areas continue to work well for weasels. A litter box must be large enough for an animal to stand within the container. Most mustelids will backup to the wall to urinate and defecate. The soiled substrate should be changed out at least daily. All will use a designated toilet area (or areas) of their choosing. Wood fuel pellets, dirt or sand are options for litterbox fill; do not use clumping cat litter (Fig. 14). Weasels generally use litterboxes as digging enrichment rather than as a latrine area.

Fig. 14 Litterbox for young minks, lined with paper towel and using wood pellets for fill.
Cage curtains are helpful to maintain the instinct for cover and to reduce stress. Sheer curtains, perhaps donated or from a resale shop, mimic the screening they would have by vegetation in a natural environment: hidden but still aware of what is going on outside their enclosure, while allowing natural lighting. Sheer cage curtains also provide the caretakers with a visualization of activity levels and what the animals are doing, or where they are, without actually opening up the curtain.
There should be plenty of cover inside the enclosure, such as dens, hide areas, branches and large pieces of fleece or fur (e.g., from cuddlecoats.com) for burrowing, enabling the animals to stay hidden, or quickly hide, while still providing opportunities for activity. The goal is to rarely see the animals while allowing opportunities for them to feel protected and safe while playing, exploring, eating and resting.
Enrichment. Juvenile enrichment should focus on three forms of enrichment important for mustelids: exploratory, ranging and predation (Vargas & Anderson 1999; Dallaire & Mason 2016; Polanco et al. 2018). These forms of play offer opportunities to learn essential survival skills.
Exploratory enrichments are the “discover and learn” enrichment components, and include a variety of toys and tunnels, surfaces with different textures, widths and diameters, moving surfaces such as suspended platforms or branches, and water features. Large muscle development is encouraged through ranging enrichment items, which include shelves, space to run under brush or other cover, climbing structures, dig boxes with dirt, leaves or other natural materials, and running wheels. Predation skills develop coordination, and include opportunities to hunt for hidden items and to chase moving objects. Random feeding times and locations are also key enrichment opportunities and can encompass all three components.
Mustelids typically spend the summer, from the time they emerge from their dens until they disperse, exploring their environments and learning to hunt under the supervision of their mother. While we cannot duplicate their natural experiences, offering a variety of rotating enrichment items can provide similar learning experiences and preparation for survival in their natural environment.
Bedding and enrichment items for young juveniles housed indoors, sized for the species, should mimic the enrichment opportunities they would experience when first emerging from the den. This can include dried leaves, pinecones, sticks, shells and small rocks; tunnels made from hollow logs, cardboard tubes (toilet paper, paper towel, printer paper, shipping tubes), PVC or small animal fabric tunnels; toys intended for small dogs, cats and ferrets such as soft toys, hide-n-seek plush and items on a fleece bungie; exercise wheels (sugar glider wheels for weasels; cat wheels for minks and fishers, if there is sufficient space); hammocks, suspended tunnels, sleep sacks, fleece honeycombs or cubes, fur pieces, pieces of flat fleece stacked up or in a pile, leafy or conifer branches, etc., can all be used and rotated for variety. Use the available air space as well as floors and walls, with furnishings such as tunnels, hammocks, honeycombs, cubes and shelves hanging from the sides and ceiling of the cage (Fig. 15). Soft toys attached to a fleece covered bungee (swivel clips on both ends) and attached to the cage wall or ceiling provide an object that will move unexpectedly when tugged and “put up a fight.” Whole prey can be suspended from or nailed to a surface. If the juveniles have access to a large “play space,” a cat wand with a soft toy on the end can give a good workout, as will a cat tree or other climbing and adventure structures.

Fig. 15 Two double Critter Nation cages joined to make an indoor enclosure for young juvenile weasels. This enclosure would typically have closed cage curtains.
Running wheels are important for both large muscle development and stress relief. Stress can trigger the “fight or flight” response, and the running wheel offers an opportunity for “flight.” The running wheel should be considered a critical component of indoor housing for weasels.
Water access
Minks require access to water for swimming once their guard hairs have grown sufficiently to provide some measure of water repellency (at around 8 weeks of age). They are naturally drawn to water and should be allowed to explore it on their own: no need to put young minks in a tub of water to teach swimming, which would be very stressful. In the wild the kits would be exploring a shoreline, pools or puddles under their mother’s watchful eyes, and dabbling in shallow water much like a young human child. Initially a small, shallow, heavy container such as a glass 9 × 13 baking pan half filled with water, or a plant saucer large enough to hold the animal without tipping over, along with some enrichment items in the water (shells, pebbles, dive toys), can be offered (Fig. 16).

Fig. 16 Young minks engaged in water play.
As minks grow, becoming better water-proofed, stronger and more coordinated and more adventurous, shallow plastic totes or tubs that they can easily climb into and out of can be placed in their enclosure and filled with water. Providing ramps for easy access in and out of deeper containers will allow gradual exploration and facilitate using the pool more quickly. As the minks become more comfortable they will readily hop in and out from all sides, while continuing to enjoy the ramp access. A variety of water containers may be offered, or swapped out, and the water should be changed frequently. Much of the water will be regularly displaced during their water play activity, and sufficient easily changeable absorbent surfaces, such as towels or thick pads, should be placed in the water activity area.
All mustelids may be provided with a pool of water and items to retrieve from the water, but it is a critical developmental component for minks. Include items that sink and also ones that float—small rocks, shells, pinecones, pool dive toys, plastic toys, bath toys, metal spoons, etc. Weasels enjoy chasing ice cubes in a shallow dish and splashing in shallow containers, such as large reptile water dishes. Provide minks with plenty of absorbent surfaces surrounding the pool to catch splashes and for drying themselves off, as their fur is only semi-waterproof. They do fine with towels, burlap sacks or bath mats while indoors. If they have access to a secure room for “playtime” prior to being moved to an outdoor enclosure, a larger tub, such as a portable stock water tank, large aquarium or large mixing tub from a home improvement store, makes a nice pool for minks. When given access to larger, deeper pools (or buckets or other unusually shaped containers) of water, minks should be supervised carefully to ensure they can easily go in and out and are comfortable with swimming. Water toys should be monitored to ensure they are not an entanglement risk. Minks love to swim under and through obstacles, rings, surface structures and tunnels, but have become trapped or been unable to get a ring off. Avoid ANY items with an opening large enough for the animal to push their head through but not large enough to easily move through entirely, which means at least a 4” diameter opening for juvenile minks. Nobody wants to be trying to remove a ring from the torso of a panicked young mink or have a mink drown because it was trapped.
Do not leave filter units in the tank unless completely secured from the enclosure occupants, as the filter unit will be dismantled and probably removed.
Minnows are both enrichment and food for minks, while providing opportunities to practice hunting skills (Fig. 17). Minks seem to instinctively know to kill a minnow by crushing the head, effectively immobilizing it. Dead, uneaten minnows (often neatly lined up) can be removed and frozen for later feeding. Large, slow minnows or suckers are easier prey than the small, fast ones. Minnows that are not quickly killed should be provided with food and hiding places, and protected from high temperatures.

Fig. 17 Minnows caught by a young mink and neatly lined up.
Juvenile care—outside enclosure
In Part 3 – Pre-Release Conditioning and Release, protocols and strategies are provided for setting up outdoor enclosures, pre-release conditioning, release and post-release support. In brief, the same principals continue to apply, with more space and less human interaction. A review of the natural history and the location where the animal came from can be used to inform the enclosure space and release site selection. The AZA Mustelid (Mustelidae) Care Manual (AZA 2010c) provides housing suggestions than can be adapted to the rehabilitation setting to reduce stress and maximize welfare while in care. The following is provided as a summary guidance.
Feeding
Continue to feed as before, although feeding times can be less frequent (two to three times per day). Increase their exposure to the type of prey they will be hunting after release, and offer at least occasional unopened whole prey. Fresh water should always be available. Ideally occasional prey species from the area around the enclosure can be live-trapped and provided to the young mustelids. This strategy offers live prey training, but equally important is the fact that constant exposure to inaccessible live prey (such as prey species running around outside the enclosure) may condition the mustelid to ignore a potential food source that would be available after release.
Housing
To reduce stress, avoid abrupt transitions, transferring as much of the previous bedding, hide-a-ways and enrichment to the new enclosure as possible. Additional enrichment ideas for the larger space meeting the exploratory, ranging and predation development needs should include a cat wheel for minks or fishers and sugar glider wheels for weasels; cat trees, shelves and platforms, branches and logs for climbing and resting; tunnels; pools of water, which for minks should be large and deep enough to allow swimming and diving and bouncy surfaces like branches, hammocks, mini trampolines and saucer swings, in addition to the items listed previously. Exercise wheels are highly valued by mustelids (Hansen & Jensen 2006), and they use them frequently, especially at night. The AZA Mustelid (Mustelidae) Care Manual (AZA 2010c) provides additional suggestions for enclosures and enrichment, which can be modified to fit the smaller spaces generally available in a rehabilitation setting.
Pre-release conditioning
Hunting behaviour generally consists of a zigzag foraging pattern in places where prey is likely to be found. Mustelids hunt individually. They do not lie and wait to ambush prey, and they do not often chase prey for long distances. They hunt using their keen sense of hearing and smell, and, once spotted, also track their prey visually. Their long, lean shape allows the weasels, minks and fishers to forage in areas not generally frequented by other predators, including under bushes, within brushy areas and windfalls, in hollow trees and logs, among the branches of trees and bushes, in piles of forest litter and in tunnels under snow, under vegetation and underground. Minks will also run along the water’s edge, peering into the water and watching for movement of aquatic prey, then diving in after it. Providing habitat and opportunities for them to engage in this exploratory behaviour also reinforces their need to stay undercover, as protection against larger predators.
Live prey training is essential. In addition to practicing their kill technique they should have the opportunity to hunt prey, and the live prey should have opportunities to hide and evade capture.
Release
The orphans raised in a rehabilitation setting do not have the months of training the wild-raised receive, nor are they familiar with the available shelters, prey or dangers encountered at the release site. Combined with the mustelid’s high metabolism, it is important to mitigate these barriers with supplemental feeding for a period of time after release, and in such a way that the target animals are getting the supplied food versus other opportunistic area wildlife. Monitoring the release site with a trail camera can be highly beneficial.
In every phase of the rehabilitation process, recognize the fact that mustelids are highly specialized, high metabolism, efficient predators with an exceptionally large range for their size and an acute sensitivity for their surroundings. This knowledge informs the decision to treat them while in care as a high stress, high activity animal with a need for a generous diet based on whole prey. An understanding of the natural history, physical characteristics and natural behaviours leads to a better understanding of their welfare and requirements while in care, and improved outcomes for the mustelids brought into rehabilitation.
Acknowledgments
The author would like to thank Dr. Roger Powell, Dr. Nicki Rosenhagen, Jessica Kainz, Cassandra Tompkins and Elliot Lozano for valuable information, comments and suggestions, and all who have shared anecdotes, photographs and feedback on the protocols.
All photos are taken by, and property of, the author, except as otherwise credited.
References
| Abou-Madi N. 2019. Natural history and medical management of mustelids. In S.M. Hernandez, H.W. Barron, E.A. Miller, R.F. Aguilar & M.J. Yabsley (eds.): Medical management of wildlife species: A guide for practitioners. Pp. 283–296. Hoboken, NJ: John Wiley & Sons, Inc. |
| Agüero M., Monne I., Sánchez A., Zecchin B., Fusaro A., Ruano M.J., del Valle A. Manuel, F.-A. R., Souto A.M., Tordable P., Cañás J., Bonfante F., Giussani E. & Terregino Calogero, O.J.J. 2023. Highly pathogenic avian influenza A(H5N1) virus infection in farmed minks, Spain, October 2022. Euro Surveillence 28(3), pii=2300001. https://doi.org/10.2807/1560-7917.ES.2023.28.3.2300001 |
| Aubry K., Wisely S., Raley C. & Buskirk, S. 2005. Zoogeography, spacing patterns, and dispersal in fishers. In D.J. Harrison, A.K. Fuller & G. Proulx (eds.): Martens and Fishers (Martes) in human-altered environments. Pp. 201–220. Boston, MA: Springer. |
| AZA Small Carnivore Taxon Advisory Group. 2010a. Reproduction- assisted rearing. In L. Byrnes, C. Dorsey & F.Spector (eds.): Mustelid (Mustelidae) care manual. Pp. 63–70. Silver Spring, MD: Association of Zoos and Aquariums. |
| AZA Small Carnivore Taxon Advisory Group. 2010b. Nutrition. In L. Byrnes, C. Dorsey & F.Spector (eds.): Mustelid (Mustelidae) care manual. Silver Spring, MD: Association of Zoos and Aquariums. Pp. 26–38. |
| AZA Small Carnivore Taxon Advisory Group. 2010c. Habitat design and containment- space and complexity. In L. Byrnes, C. Dorsey & F.Spector (eds.): Mustelid (Mustelidae) care manual. Silver Spring, MD: Association of Zoos and Aquariums. Pp. 14–18. |
| Blus, L.J., Henny, C.J. & B.M. Mulhern. 1987. Concentrations of metals in mink and other mammals from Washington and Idaho. Environmental Pollution 44(4), 307–318, doi: 10.1016/0269-7491(87)90206-5 |
| Dallaire J.A. & Mason G. 2016. Play in juvenile mink: litter effects, stability over time, and motivational heterogeneity. Developmental Psychobiology 58 945–957, doi: 10.1002/dev.21425 |
| Devaux C.A., Pinault L., Delerce J., Raoult D., Levasseur A. & Frutos R. 2021. Spread of mink SARS-CoV-2 variants in humans: a model of sarbecovirus interspecies evolution. Frontiers in Microbiology 12, 675528, doi: 10.3389/fmicb.2021.675528 |
| Frost H.C. & Krohn, W.B. 1994. Capture, care, and handling of fishers (Mattes pennanti). Maine Agricultural and Forest Experiment Station Technical Bulletin 157. Accessed on the internet at https://digitalcommons.library.umaine.edu/aes_techbulletin/57/ on 21 May 2021. |
| Hall E.R. 2015. American weasels. P. 88. Lexington, KY: Jefferson Publications. |
| Hansen S.W. & Jensen, M.B. 2006. Quantitative evaluation of the motivation to access a running wheel or a water bath in farm mink. Applied Animal Behaviour Science 98(1–2), 127–144, doi: 10.1016/j.applanim.2005.08.017 |
| Heidt G.A. 1970. The least weasel, Mustela nivalis Linnaeus, developmental biology in comparison with other North American Mustela. Biological Series 4(7) 237. |
| Hornshaw T.C., Aulerich R.J. & Johnson H.E. 1983. Feeding great lakes fish to mink: effects on mink and accumulation and elimination of PCBS by mink. Journal of Toxicology and Environmental Health 11(4–6), 933–946, doi: 10.1080/15287398309530396 |
| King C.A. & Powell R.A. 2007a. Reproduction. In The natural history of weasels and stoats: Ecology, behavior, and management (2nd edn.). Pp.199–229. New York, NY: Oxford University Press. |
| King C.A. & Powell R.A. 2007b. Hunting behavior. In The natural history of weasels and stoats: ecology, behavior, and management (2nd edn.). Pp. 113–115. New York, NY: Oxford University Press. |
| Kwon D., Lee W., Kim S.H. & Jung, Y.S. 2022. Comparison of hepatic metabolite profiles between infant and adult male mice using 1H-NMR-based untargeted -metabolomics. Metabolites 12(10), 910, doi: 10.3390/metabo12100910 |
| Ljungvall K., Magnusson U., Korvela M., Norrby M., Bergquist J. & Persson S. 2017. Heavy metal concentrations in female wild mink (Neovison vison) in Sweden: sources of variation and associations with internal organ weights. Environ Toxicol Chem 36(8), 2030–2035, doi: 10.1002/etc.3717 |
| Luxon M., Toll J. & Hanson C. 2014. Assessing effects of PCB exposure on American mink (Mustela vison) abundance in Portland Harbor. Integrated Environmental Assessment and Management. 10(1), 60–68, doi: 10.1002/ieam.1498. |
| Mayer J., Marini R.P. & Fox, J.G. 2015. Biology and -diseases of Ferrets. In J.G. Fox, C.L. Anderson, G.M. Otto, K.R. Pritchett-Corning & M.T. Whary (eds.): Laboratory -animal medicine (3rd edn). Pp. 577–622. Cambridge, MA: Academic Press. |
| Molenaar R.J., Vreman S., Hakze-van der Honing R.W., Zwart R., de Rond J., Weesendorp E., Smit L.A.M., Koopmans R.B., Stegeman A. & van der Poel, W.H.M. 2020. Clinical and pathological findings in SARS-CoV-2 disease outbreaks in farmed mink (Neovison vison). Veterinary Pathology 57(5), 653–657, doi: 10.1177/0300985820943535 |
| Polanco A., Díez-Leon A. & Mason G. 2018. Stereotypic behaviours are heterogeneous in their triggers and treatments in the American mink, Neovison vison, a model -carnivore. Animal Behaviour 141, 105–114, doi: 10.1016/j.anbehav.2018.05.006 |
| Popp P. (2024). Weasels, minks, and fishers—Orphan rehabilitation of the slinky carnivores, Part 1: natural history. Wildlife Rehabilitation Bulletin 42(2) 40–49, doi: 10.53607/wrb.v42.278 |
| St-Pierre P., Pilon G., Dumais V., Dion C., Dubois M.J., Dubé P, Desjardins Y. & Marette, A. 2014. Comparative analysis of maple syrup to other natural sweeteners and evaluation of their metabolic responses in healthy rats. Journal of Functional Foods 11, 460–471, doi: 10.1016/j.jff.2014.10.001 |
| Taylor D.R. 2014. The Ferret in viral respiratory disease research. In J.G. Fox & R.P. Marini (eds.): Biology and -diseases of the Ferret (3rd edn.). Pp. 629–640. Ames, IA: John Wiley & Sons, Inc. |
| Vargas A. & Anderson S.H. 1999. Effects of experience and cage enrichment on predatory skills of black-footed ferrets (Mustela nigripes). Journal of Mammalogy 80, 263–269, doi: 10.2307/1383226 |
| Zhou P. & Shi Z-L. 2021. SARS-CoV-2 spillover events. Science. 371(6525), 120–122, doi: 10.1126/science.abf6097 |